Healthcare facilities collect data on healthcare-associated infections (HAIs) using standardized definitions and may share data with different groups inside (e.g., unit staff, Infection Control Committee) and outside the facility (e.g., Board of Directors, health department, general public). HAI data are used for a variety of purposes, which may include satisfying reporting mandates, comparing infection rates between and within healthcare facilities, providing consumers with information, guiding policies and procedures, evaluating the effectiveness of interventions, and conducting research.
HAI Data Uses
Detect Trends
Identify Risk Factors
Guide Treatment
Assess Prevention Impact
Detect Clusters & Outbreaks
Surveillance Data can be Categorized into Process Measures or Outcome Measures
Process Measures
Measures a process that may affect outcomes
Process measures have a 100% target rate and are less complicated because they do not require risk adjustment. These measures apply to a variety of healthcare settings and involve direct care / provider accountability.
Examples: Hand hygiene, Surgical Care Improvement Project (SCIP) measures
Outcome Measures
Measures actual results
Outcome measures have variable goals and often require risk adjustment, making them more complicated. These measures may not be collected in all healthcare settings and may not involve direct care / provider accountability.
Examples: CLABSI, SSI, CAUTI
Common HAI Outcome Measures
Purpose:
- Probability or risk of an infection in a population
Calculation:

Interpretation:
- Rate usually expressed per 1,000 device days (if device-associated) or per 100 procedures (if procedure-associated)
- Not risk-adjusted
Purpose:
- A measure of device use (relative to the number of patient days)
Calculation:

Interpretation:
- Similar to a rate
- Usually expressed as a percent
- Not risk-adjusted
Purpose:
- A risk-adjusted summary measure used to track device use at a national, state, or local, or facility level over time
Calculation:

Interpretation:
- If the SUR <1.0, fewer device days were reported than predicted.
- If the SUR = 1.0, the same number of device days were observed as predicted.
- If the SUR > 1.0, more device days were observed than predicted.
- The SUR is designed to be a high-level indicator of device use, and should not be used to draw conclusions around whether devices are overused or underused.
- The SUR should be used in conjunction with the SIR.
Purpose:
- A risk-adjusted summary measure used to track HAIs at a national, state, or local level over time
Calculation:

Interpretation:
- If the SIR <1.0, fewer infections were observed than predicted.
- If the SIR = 1.0, the same number of infections were observed as predicted.
- If the SIR >1.0, more infections were observed than predicted.
Purpose:
- A risk-adjusted summary measure that indicates the number of infections (e.g., CLABSI, CAUTI, C. difficile) that must be prevented within a group, facility, or unit to achieve an HAI reduction goal
Calculation:

Interpretation:
- A positive CAD is the number of excess infections a facility would have needed to prevent to achieve an HAI reduction goal during a specified time period.
- A negative CAD means the facility has reached the HAI reduction goal.
- Usually presented as a whole number.
Purpose:
- A standardized metric of antimicrobial use for specified patient care locations
Calculation:
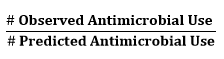
Interpretation:
- If the SAAR <1.0, less antimicrobial use was observed than predicted.
- If the SAAR = 1.0, the same antimicrobial use was observed as predicted.
- If the SAAR >1.0, more antimicrobial use was observed than predicted.
- Not a definitive measure of appropriateness or judiciousness of antimicrobial use and any SAAR value may warrant additional investigation.
Opens pdf to download
Opens document to download
Opens in a new window
External link will open in new window. Click link to exit Virginia Department of Health Website.
