No Emergency Alerts at This Time
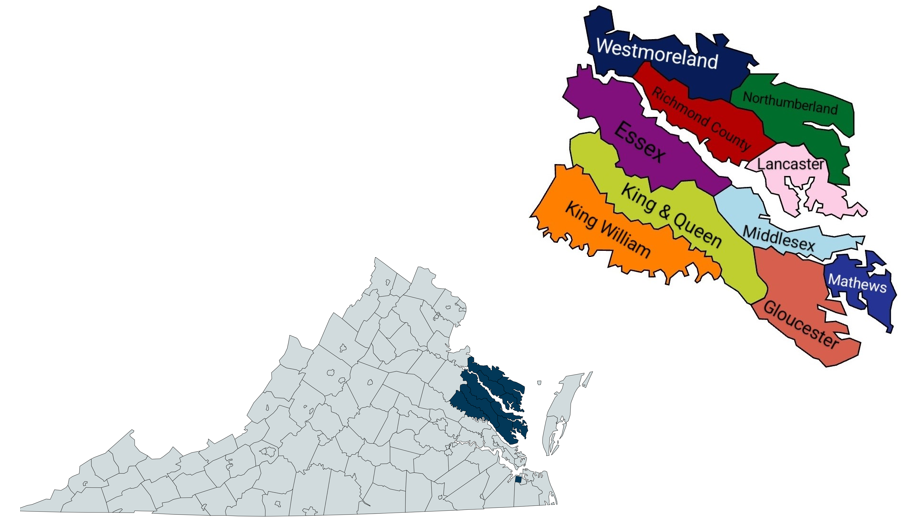
THE THREE RIVERS HEALTH DISTRICT is the 10 county Virginia Department of Health district for Virginia’s beautiful Northern Neck and Middle Peninsula. This 2,000 square mile area is located between the waters of the Potomac, Rappahannock, and York Rivers and borders the Chesapeake Bay on the east. Our public health professionals serve a population of approximately 140,000, including 3 Native American reservations, 9 incorporated towns, and a large number of visitors.