
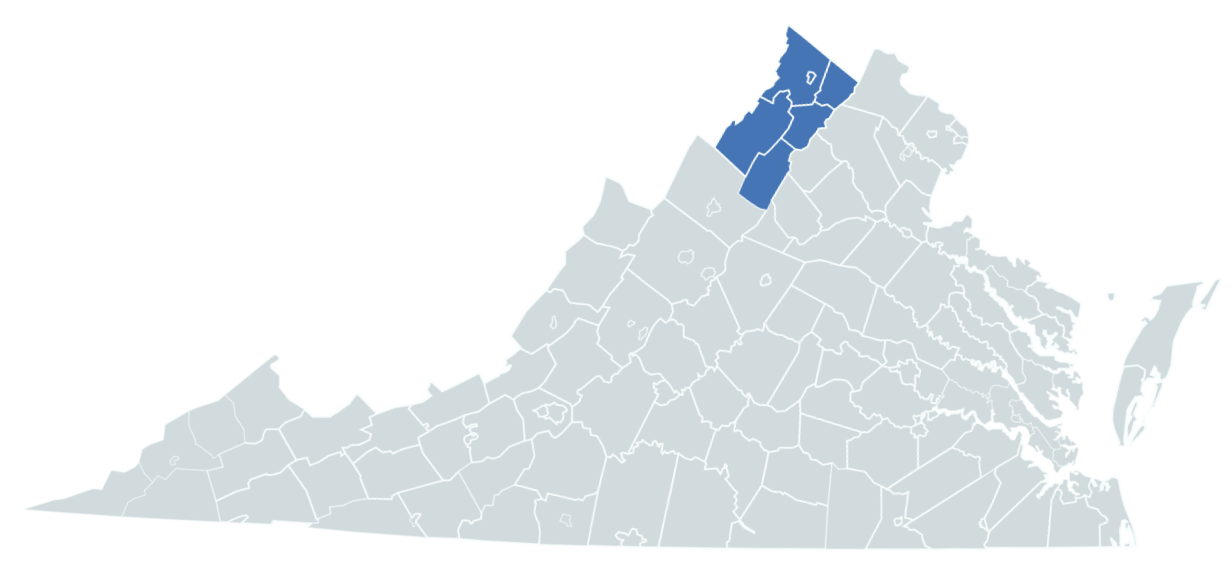
Serving the City of Winchester and the counties of Clarke, Frederick, Page, Shenandoah, and Warren.
** PLEASE NOTE - The Lord Fairfax Health District serving Clarke, Frederick, Page, Shenandoah and Warren County and the city of Winchester are closed Friday June 12, 2026 for staff training purposes. We will reopen at 0800 Monday June 15, 2026. We apologize for any inconvenience. **
Lord Fairfax Health District operates 5 local health departments within the counties of Clarke, Frederick, Page, Shenandoah, Warren, and the City of Winchester. Striving to improve the health of its community members, the Lord Fairfax Health District provides many services to protect and promote the health and well-being of the health of individuals and the environment.
Our Mission: Through partnerships and collaboration, LFHD promotes the health and well-being of our communities through education, prevention, and access to quality public health services.
Our Vision: LFHD works alongside the communities we serve to create a safe, healthy, and equitable environment for all.
Health Director: Tara Blackley MA, MPH, MBA
Medical Officer: Dr. Richard Williams, MD
Questions? AskLFHD@vdh.virginia.gov
Find us on Facebook!
Find Local Resources
Need help? Search free and reduced-cost services like medical care, food, housing, and more in your area.
Frederick / Winchester
Health Department
10 Baker St.
Winchester, VA 22601
Phone: (540) 722-3470
Fax: (540) 722-3475
Frederick / Winchester
Environmental Health
107 North Kent St., Suite 201
Winchester, VA 22601
Phone: (540) 722-3480
Fax: (540) 722-3479
Clarke County
Health Department
100 North Buchmarsh St.
Berryville, VA 22611
Phone: (540) 955-1033
Fax: (540) 955-4094
Warren County
Health Department
465 W. 15th St., Suite #200
Front Royal, VA 22630
Phone: (540) 635-3159
Fax: (540) 635-9698
Page County
Health Department
75 Court Lane
Luray, VA 22835
Phone: (540) 743-6528
Fax: (540) 743-3811
Shenandoah County
Health Department
494 North Main St., Suite #100
Woodstock, VA 22664
Phone: (540) 459-3733
Fax: (833) 608-4964
