Potassium Iodide (KI) is a protective, over-the counter supplement which helps to protect the thyroid gland against internal uptake of radioiodines that may be released in the unlikely event of a nuclear reactor accident. Detailed information about KI is available by clicking Potassium Iodide (KI) Fact Sheet 11Jan13.
Not all nuclear power plant accident scenarios may result in a release of radioiodine requiring the distribution and use of KI. Nuclear power plants are designed to remove certain radioactive materials during an accident using filters and sprays. However, in ceratin types of accidents this may not always be possible.
The Nuclear Regulatory Commission (NRC) provided KI to states for use in the 10 mile plume phase emergency planning zone (EPZ) of commercial nuclear power stations.
VDH will recommend distribution and use of KI for members of the public at the Evacuation Assembly Centers (EAC) when the projected radiation dose to the thyroid gland exceeds 5 rem.
KI that will be made available to emergency workers and the public is shown below, followed by lot numbers and expiration dates:
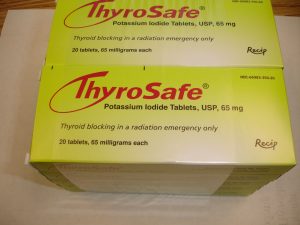

- Lot Number: TU905A
- Expiration Date: 10/31/2029
Disposal
Proper disposal of Potassium Iodide (KI) is in regular household, dumpster, or landfill garbage. DO NOT FLUSH the tablets or otherwise put them into the water supply.
Other Information about KI
U.S. Nuclear Regulatory Commission’s web page on KI:
https://www.nrc.gov/about-nrc/emerg-preparedness/about-emerg-preparedness/potassium-iodide-use.html
U.S. Food and Drug Administration’s (FDA) web site on radiation emergencies:
http://www.fda.gov/Drugs/EmergencyPreparedness/BioterrorismandDrugPreparedness/ucm063807.htm
FDA Guidance: Potassium Iodide as a Thyroid Blocking Agent in Radiation Emergencies:
www.fda.gov/downloads/Drugs/GuidanceComplianceRegulatoryInformation/Guidances/ucm080542.pdf
Centers for Disease Control and Prevention:
http://www.bt.cdc.gov/radiation/ki.asp
Office of Radiological Health | 109 Governor Street, 7th Floor | Richmond, VA 23219
Telephone (804) 864-8150 | Fax: (804) 864-8155
